I. Introduction: Engineering the Modern Injection
In the global medical consumable supply chain, the distinction between precision instruments and standard tools dictates clinical outcomes. A frequent, foundational question in biomedical engineering and procurement is the agulha hipodérmica vs agulha normal comparison. While a “regular needle”—such as a surgical suture needle or a traditional textile needle—is a solid piece of drawn steel designed strictly to pierce and pass thread through a substrate, a hypodermic needle is an entirely different feat of engineering. The word “hypodermic” originates from Greek, meaning “under the skin.” Therefore, medical hypodermic needles are highly specialized, hollow tubes (cannulas) engineered to deliver fluids, medications, and vaccines directly into the body’s tissues or vascular system, or to extract fluids like blood for vital diagnostic testing.
As a premier manufacturer of medical specialty needles and state-of-the-art automated inspection equipment for medical production lines, our facility understands that manufacturing a hollow cannula requires exponentially more precision than a solid needle. The structural integrity of the ultrathin stainless steel wall, the microscopic geometry of the beveled tip, and the fluid dynamics of the internal lumen are critical. A microscopic burr or a misaligned bevel can cause severe patient trauma, tissue coring, or medication sheer.
Therefore, our role among global hypodermic needle manufacturers goes far beyond basic metalworking. We provide a comprehensive portfolio of injection tools engineered with zero-defect tolerances. We cater strictly to B2B procurement networks, hospital systems, and clinical distributors, ensuring that our supply chain partners receive instruments that perform flawlessly under high clinical pressure. This guide serves as a definitive technical portfolio, detailing our manufacturing capabilities, the precise specifications of various gauges, and our unwavering commitment to automated quality control.
II. Anatomy & Connection Standards
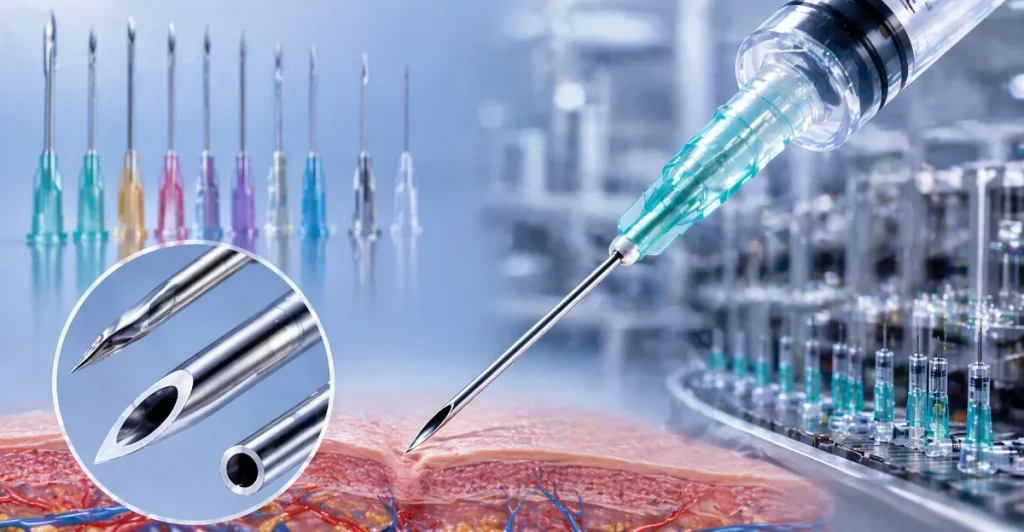
To evaluate our product lines effectively, procurement officers and clinical directors must understand the structural anatomy of our needles. We do not just assemble parts; we engineer every microscopic element of the needle to optimize the flow rate and minimize patient discomfort.
A. The Bevel and Lumen Physics
The performance of any hypodermic needle relies heavily on two elements: the bevel (the slanted tip) and the lumen (the hollow interior).
- The Bevel: We utilize advanced, automated multi-angle grinding machinery to create standard, short, and true intradermal bevels. The primary angle cuts the initial path, while the secondary angles part the tissue smoothly rather than tearing it. This multi-beveled approach requires constant automated optical inspection to ensure absolute sharpness.
- The Lumen: The inner diameter determines the fluid dynamics. Through proprietary thin-wall and ultra-thin-wall extrusion techniques, we maximize the lumen size without compromising the outer diameter of the cannula, allowing for faster flow rates of highly viscous medications (like testosterone or concentrated biologics) even through smaller gauge needles.
B. Hub Designs and Connections
A needle is only as secure as its connection to the syringe. The hub, typically manufactured from medical-grade polypropylene, must lock seamlessly to prevent blowout under high-pressure injections.
- Luer Lock Technology: O nosso luer lock hypodermic needles feature a threaded hub design that twists and securely locks onto the corresponding threads of a luer lock syringe. This is an absolute necessity for administering viscous medications, drawing thick blood, or connecting to automated IV infusion pumps where pressure variations could dislodge a standard slip-fit needle.
- Color-Coding: All hubs are strictly color-coded according to ISO 6009 standards, allowing fast, visual identification of the needle gauge by busy clinical staff in high-stress environments.
C. Integrated Delivery Systems
While we are a primary supplier of standalone needles, we also manufacture and package complete, integrated systems. Distributors can procure bulk quantities of matching agulhas e seringas hipodérmicas, or opt for a pre-assembled, blister-packed hypodermic syringe with needle to streamline the clinical workflow, reduce packaging waste, and minimize the risk of contamination during assembly.
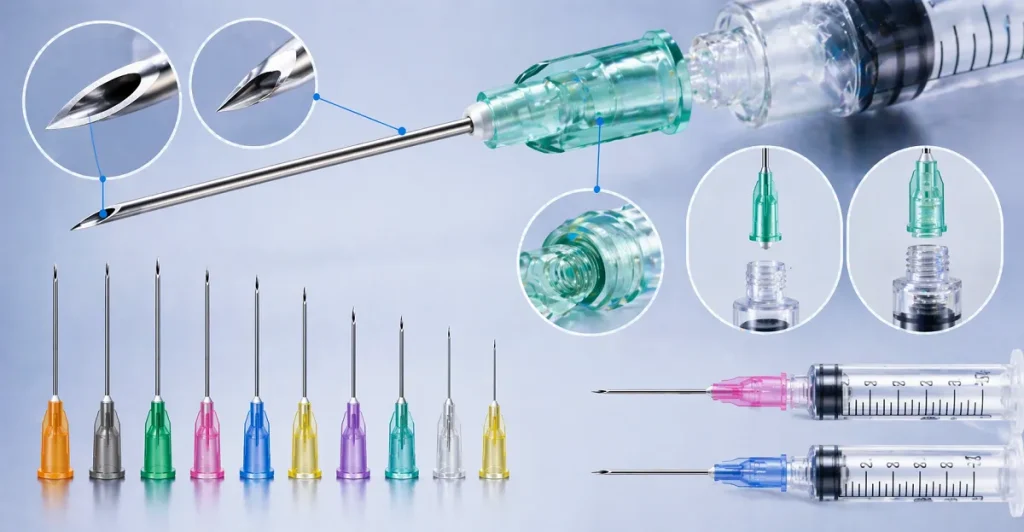
III. Product Portfolio: Classifying Hypodermic Needle Types by Size
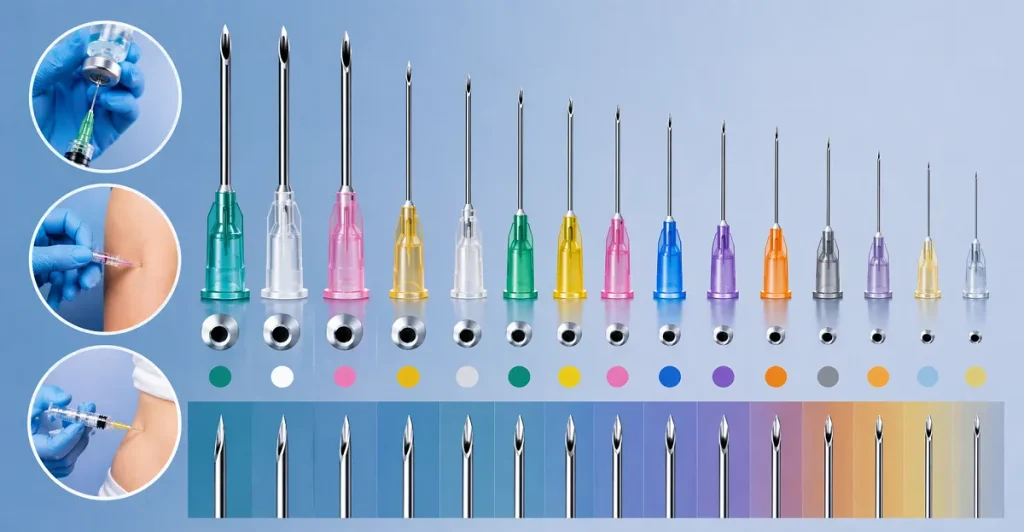
The Birmingham Wire Gauge (BWG) system is the universal standard for needle sizing. It operates on an inverse scale: the lower the gauge number, the larger the outer diameter of the needle. Our manufacturing lines are capable of producing a vast array of hypodermic needle types, meeting the exact hypodermic needle dimensions required for any specific clinical, cosmetic, or veterinary application.
Below is a detailed breakdown of our portfolio, categorized by hypodermic needle sizes and intended applications, highlighting the exact hypodermic needle diameter for clinical procurement accuracy.
A. The Large Gauge Series (Trauma, Blood Banking, and Veterinary)
Large gauge needles require immense structural integrity. They are designed to penetrate tough dermal layers and facilitate the rapid movement of high-volume or highly viscous fluids. Our large gauge hypodermic needle products are drawn from premium 304 stainless steel to ensure they will never buckle under force.
- 7 Gauge and 12 Gauge: These represent the extreme end of the spectrum. A 7 gauge hypodermic needle (approx. 4.5mm outer diameter) and a 12 gauge hypodermic needle (approx. 2.7mm outer diameter) are rarely used in standard human medicine. They are primarily utilized as veterinary hypodermic needles for large livestock (equine, bovine) to administer thick antibiotic slurries, or in specialized biopsy/bone marrow extraction instruments.
- 14 Gauge and 16 Gauge: O 14 gauge hypodermic needle (approx. 2.1mm OD, typically with a pale green hub) and the 16g hypodermic needle (approx. 1.6mm OD, typically with a white hub) are critical life-saving tools. They are the standard for hospital trauma bays and emergency departments requiring rapid fluid resuscitation or emergency blood transfusions. Additionally, the 16G is the global standard for blood bank collection lines, as the wide lumen prevents the shearing and destruction of red blood cells during high-volume donation.
B. The Standard Clinical Series (Phlebotomy and General Injection)
These gauges are the high-volume workhorses of hospitals, laboratories, and outpatient clinics globally.
- 18 Gauge and 20 Gauge: O 18 gauge hypodermic needle (pink hub, 1.2mm OD) is the universal standard for medication preparation. Nurses utilize these to draw thick medications from vials or to mix intravenous compounds. The 20 gauge hypodermic needle (yellow hub, 0.9mm OD) bridges the gap between prep and patient care, often used for minor surgical procedures, IV catheter placement, and drawing blood from healthy, robust veins.
- 21 Gauge, 23 Gauge, and 24 Gauge: This cluster represents routine patient care. The 21g hypodermic needle (green hub, 0.8mm OD) is the most common needle used for routine phlebotomy and blood draws. The 23g hypodermic needle (blue hub, 0.6mm OD) is the global standard for intramuscular (IM) injections, such as influenza and COVID-19 vaccines, deep into the deltoid muscle. For patients with smaller or more fragile veins, the 24 gauge hypodermic needle (purple hub) provides a perfect balance of fluid flow and patient comfort.
C. The Fine Gauge Series (Subcutaneous, Cosmetic, and Diabetic)
Engineered with ultra-thin walls and advanced micro-silicone coatings, our fine gauge series prioritizes a painless patient experience for shallow injections.
- 25 Gauge and 27 Gauge: O 25g hypodermic needle (orange hub, 0.5mm OD) is widely used for subcutaneous injections and local anesthetics. The 27 gauge hypodermic needle (grey hub, 0.4mm OD) is a staple in dental clinics for nerve blocks, in cosmetic dermatology for Botox and dermal filler injections, and in pediatrics.
- 28 Gauge and 30 Gauge: These are our micro-cannulas. The 28 gauge hypodermic needle and the ultra-fine 30g hypodermic needle (yellow/clear hubs, 0.3mm OD) are specifically manufactured as a hypodermic needle insulin delivery solution. Diabetic patients require daily, often multiple, injections. By matching the precision and comfort of premium consumer brands like easy touch hypodermic needles, our OEM products ensure pain-free subcutaneous penetration without tissue tearing.
Authoritative Reference Table: Standard Hypodermic Needle Specifications
| Gauge | Hub Color Standard (ISO 6009) | Nominal Outer Diameter (mm) | Aplicação clínica primária |
| 12G | No Standard (Often White/Clear) | 2.77 mm | Veterinary, Specialized Biopsy |
| 14G | Pale Green | 2.10 mm | Trauma Resuscitation, Central Lines |
| 16G | Branco | 1.65 mm | Blood Banking, Dialysis |
| 18G | Cor-de-rosa | 1.27 mm | Medication Preparation, Fluid Transfer |
| 20G | Amarelo | 0,90 mm | IV Catheter Insertion, Venipuncture |
| 21G | Verde profundo | 0.81 mm | Standard Phlebotomy / Blood Draw |
| 23G | Light Blue | 0.64 mm | Intramuscular (IM) Vaccines |
| 25G | Laranja | 0.515 mm | Subcutaneous Injections, Local Anesthesia |
| 27G | Cinzento | 0.41 mm | Cosmetic Injectables, Dental Blocks |
| 30G | Yellow (Diabetic Std) | 0.31 mm | Insulin Delivery, Allergy Testing |
(Note: Hub colors follow ISO 6009 where applicable, but diabetic and specialty veterinary lines may utilize independent color-coding systems based on regional distributor preferences.)
IV. Safety Innovations and Clinical Compliance
As global healthcare networks tighten their regulatory standards regarding bloodborne pathogens, the demand for advanced safety engineering has become paramount. Supplying a sharp piece of steel is no longer sufficient; the modern manufacturer must provide integrated risk-mitigation solutions.
A. Single-Use Mandates and Sterilization
Every product leaving our facility is certified as a strictly single-use, disposable hypodermic needles unit. The reuse of needles is a catastrophic clinical failure resulting in the transmission of HIV, Hepatitis B, and Hepatitis C. We guarantee a perfectly sterile hypodermic needle through automated, high-volume Ethylene Oxide (EtO) gas sterilization chambers or Gamma Irradiation, coupled with medical-grade blister packaging using Tyvek materials to ensure absolute barrier integrity for a minimum of 5 years.
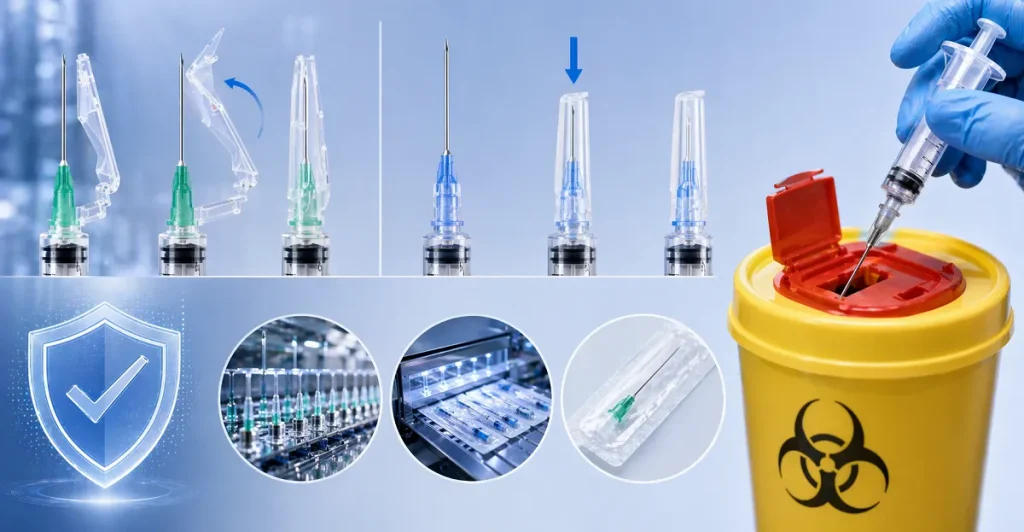
B. Engineered Sharps Injury Protection (ESIP)
Accidental needlesticks are a major occupational hazard for nurses and doctors. To combat this, we manufacture a comprehensive line of safety hypodermic needle products.
- Hinged Safety Shields: We produce needles with an integrated, hinged plastic arm that the clinician can snap over the needle using a single hand immediately after injection.
- Retractable Mechanisms: For higher-tier procurement, we offer spring-loaded retractable syringes where the needle pulls back entirely into the syringe barrel post-injection. Our safety mechanisms are engineered to meet or exceed the performance benchmarks set by legacy safety products, such as the magellan hypodermic safety needle, ensuring that our B2B clients can offer top-tier safety compliance to their hospital networks.
C. Post-Use Protocols
While we design our needles to be incredibly safe prior to and immediately after use, we strongly advocate for rigorous hypodermic needle disposal protocols. All clinical facilities must deposit used needles directly into FDA-cleared, puncture-proof sharps containers to ensure the absolute safety of biomedical waste disposal teams.
V. B2B Sourcing and Automated Manufacturing Excellence
For global medical networks evaluating hypodermic needle suppliers, the decision matrix extends beyond the physical product; it encompasses the reliability, scalability, and quality control of the manufacturing partner.
What separates us from generic market suppliers is our foundation in building the very machinery that makes the consumables. Because we are experts in automated inspection equipment for medical production lines, our needle manufacturing is subjected to rigorous, inline, high-speed optical sorting that human inspectors cannot match.
- Precision Metallurgy: Our process begins with continuous strips of surgical-grade 304 or 316L stainless steel. The strip is rolled into a precise tube and TIG welded in an inert argon atmosphere to prevent oxidation.
- Cold Drawing: The tube is drawn repeatedly through carbide dies, systematically reducing its diameter and wall thickness to achieve exact gauge dimensions while aligning the steel’s grain structure for maximum tensile strength.
- Automated Bevel Grinding: Utilizing advanced CNC grinding wheels, each tip is cut at optimal primary and secondary angles. Our automated optical inspection systems instantly reject any needle with a microscopic burr, “fishhook” tip, or blunt edge.
- Siliconization: The cannulas are coated with a micro-layer of medical-grade, friction-reducing silicone. This ensures that the needle glides through the skin, minimizing pain and tissue trauma.
- Automated Assembly and Bonding: The cannula is mated to the luer hub using UV-cured medical epoxy, tested to withstand significant pull-force without detaching.
Our commitment to zero-defect manufacturing means that when a clinical distributor orders ten million units of a 21G needle, every single unit will perform identically. This is the hallmark of professional B2B medical manufacturing.

VI. Frequently Asked Questions (Q&A)
To further assist procurement officers, distribution sales representatives, and biomedical technicians, we have compiled the most frequently asked questions regarding our manufacturing processes, product specifications, and clinical usage.
A hypodermic needle is a highly specialized, hollow medical instrument designed to be attached to a syringe. The word “hypodermic” literally translates to “under the skin.” Unlike solid needles used for sewing or surgical suturing (which only pass thread through a substrate), the hollow core (known as the lumen) of a hypodermic needle allows medical professionals to inject liquid medications, vaccines, or intravenous fluids directly into the body’s tissues or vascular system. Conversely, it allows for the precise extraction of bodily fluids, such as blood, for diagnostic laboratory testing.
The difference lies in how the needle hub connects to the syringe barrel. A Luer Lock needle features small internal plastic threads inside the hub that allow it to twist and securely lock onto the threaded tip of a Luer Lock syringe. This prevents the needle from popping off during high-pressure injections or when drawing thick fluids. A Luer Slip needle simply slides and uses friction to stay on the syringe tip. For most critical medical applications, especially involving viscous drugs or vascular access, Luer Lock is the preferred standard.
The modern hypodermic needle was independently invented in 1853 by Scottish physician Alexander Wood and French surgeon Charles Pravaz. Wood developed a hollow needle attached to a plunger to inject morphine for localized pain relief, while Pravaz created a similar silver syringe mechanism. Their breakthrough innovations paved the way for the precision, mass-manufactured stainless steel medical instruments used in hospitals and clinics globally today.
The “G” stands for Gauge, specifically referring to the Birmingham Wire Gauge (BWG) system used to measure the outer diameter of medical tubing. The gauge system operates on an inverse scale: the higher the gauge number, the thinner the needle. For example, a 30G needle is an ultra-fine needle used for insulin, while a 14G needle is a very thick needle used for rapid trauma resuscitation.
The reduction in patient pain is the result of three specific manufacturing techniques. First, the needle is ground with a multi-beveled tip (usually three distinct angles) that allows it to part tissue fibers smoothly rather than tearing them. Second, the needle undergoes an electro-polishing process to remove any microscopic burrs or jagged edges. Finally, the entire cannula is coated with a micro-thin layer of medical-grade silicone lubricant, which drastically reduces friction as the needle passes through the dermal layers.
Used needles represent a significant biohazard and must never be thrown into regular household or commercial trash. Immediately after use, the needle (and the attached syringe) must be placed directly into a designated, rigid, puncture-proof FDA-cleared sharps disposal container. Once the container is filled to the indicated line, it must be sealed and disposed of according to local biomedical waste regulations, which generally involves incineration or autoclave sterilization by specialized medical waste collection services.
When manufactured, sterilized, and packaged correctly in medical-grade blister packs (typically a combination of medical paper/Tyvek and a plastic film), a disposable hypodermic needle generally has an officially certified shelf life of 5 years from the date of sterilization. This shelf life remains valid only if the packaging is not torn, punctured, water-damaged, or exposed to extreme environmental degradation. If the package is compromised, the needle is no longer considered sterile.
Hypodermic needles are manufactured through a highly automated, precision-engineering process. It begins with a flat strip of surgical-grade stainless steel that is rolled into a tube and laser-welded shut. This tube is repeatedly drawn through a die (a process called cold drawing) to stretch it and reduce its diameter to the exact desired gauge. The tubes are then cut to length, and one end is precisely ground at multiple angles to create a razor-sharp, beveled tip. Finally, they are attached to a plastic hub via epoxy, lubricated with medical-grade silicone for smooth insertion, sterilized via Ethylene Oxide gas, and sealed in blister packaging.